Preclinical:
The Research Powering IGC-AD1
IGC-AD1 is built on preclinical research conducted on its active pharmaceutical ingredients (APIs). At low concentrations, the APIs showed promising results in both in vitro and in vivo efficacy assays related to the hallmarks of Alzheimer’s disease.
Early Lab Discoveries
That Inspire Hope
Preclinical studies laid the foundation for IGC-AD1 by testing its dual active ingredients in Alzheimer’s disease cells and mice models.
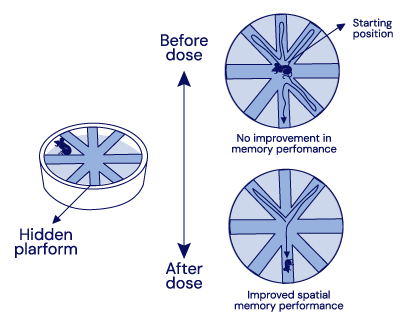
Preclinical Spatial Memory Study in APP/PS1 Transgenic Mice
APP/PS1 Transgenic mice treated with IGC-AD1 API showed about a 50% reduction in both latencies to escape and number of errors, compared to untreated controls.
Reduces Key
Alzheimer’s Features
IGC-AD1 API preserves Aβ40 Monomers and Inhibits Aggregation
as Assessed by ThT Assay.
Mitochondrial Respiratory Activity in IGC-AD1
API-Treated APP/PS1 Mouse Model
Treatment with IGC-AD1 API led to improved and restored mitochondrial function in the hippocampus and cortex of APP/PS1 Transgenic Mouse Model, two regions affected in Alzheimer’s disease.
The science behind IGC-AD1: Early indicators that matter
for Alzheimer’s Disease research
The API in IGC-AD1 reduces Aβ40 peptide production and Aβ42 aggregation in Alzheimer’s cell lines.
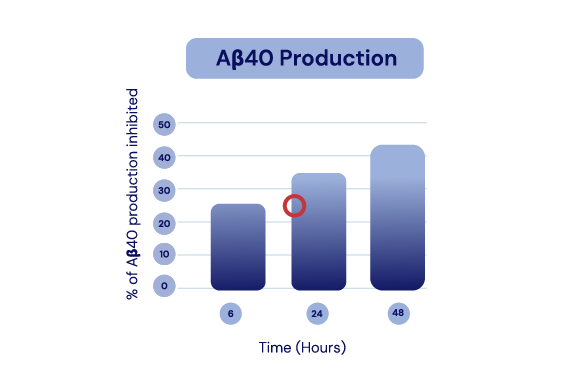

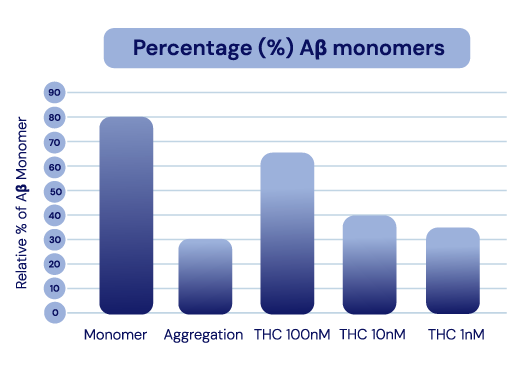
In Alzheimer’s cell lines, IGC-AD1 increased Aβ monomers and decreased Aβ aggregation in a dose-dependent manner.
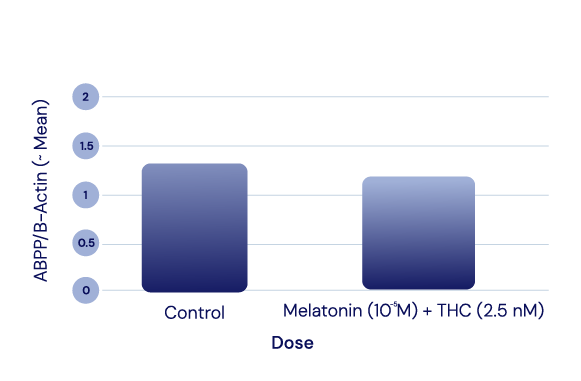
APP Levels
The APIs in IGC-AD1 did not reduce Amyloid Precursor Protein (APP) levels in Alzheimer’s cell lines. APP modulates cell growth, motility, and survival; it is cut to create small fragments such as the Aβ peptide that eventually deposit as plaque.
Spatial Memory
Spatial Memory Study in APP/PS1 Transgenic Mice In the Radial Arm Water Maze (RAWM) test, IGC-AD1 API treatment resulted in over a 50% reduction in both latencies to escape and number of errors in APP/PS1 transgenic mice, compared to untreated controls.
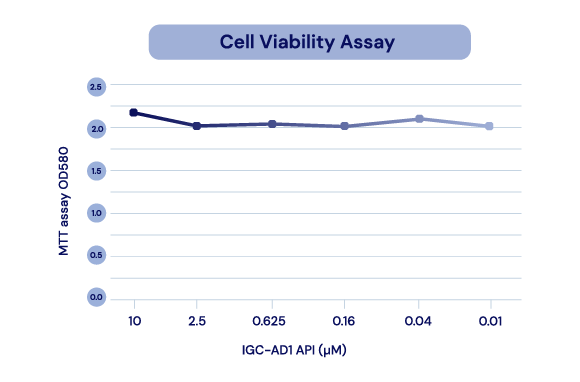
Neurotoxicity
Over 48 hours, repeated low-concentration exposure to the API in IGC-AD1 was not toxic to Alzheimer’s cells (N2a/AßPPsWe cells).
